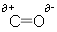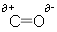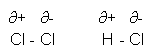Intermolecular Forces
- Energy is needed to change liquids to gases. This energy is used to overcome the forces in between the separate molecules.
- In Alkanes and Alkenes, the larger the molecule, the stronger the intermolecular forces.
- The intermolecular forces arise due to the presence of dipoles in the molecules.
- A dipole arises when a charge is not shared equally between two molecules, take for instance a carbon-oxygen bond:
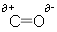
- The oxygen atom has a greater electronegativity, and so the bonding pair of electrons lies closer to the oxygen atom than the carbon atom. This causes the carbon atom to have a slight positive charge, and the oxygen atom to have a slight negative charge.
- The molecule has a dipole; it is said to be polarised.
Permanent Dipoles
- These occur when two atoms bonded to each other have significantly different electronegativities.
- Hydrogen has a lower electronegativity than chlorine, and so when chlorine bonds with hydrogen, a dipole is formed.
Instantaneous Dipoles
- Atoms with similar/same electronegativities do not have permanenet dipoles; however they do have instantaneous dipoles due to the constant movement of the electrons.
Induced Dipole (for example a Br2 molecule is polarised next to a C=C bond)
- If an unpolarised molecule finds itself next to a polar molecule, the unpolarised molecule may get a dipole induced in it. This happens when hydrogen chloride comes into contact with a chlorine molecule:
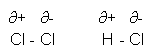
Intermolecular Forces
- All intermolecular forces arise from the attractive forces between dipoles; there are three types:
- Permanent Dipole- Permanent Dipole: Two or more permanent dipoles are attracted to each other, for example in HCl.
- Permanent Dipole- Induced Dipole: a permanent dipole induces a dipole in another molecule, this causes an attraction between the molecules, for example between HCl and Cl2.
- Instantaneous Dipole Induced Dipole (Van der Waals forces / London forces / Dispersion): where an instantaneous dipole induces a dipole in another molecule, for example between Cl2 molecules. These attractions occur between all molecules.
- To decide whether a molecule has a dipole, the electronegativities of the elements must be looked at, in addition to the shape of the molecule.
- Elemeents towards the top right of the periodic table are more electronegative than molecules to the bottom left.
- Pauling’s electronegativity values are shown below:

Permanent Dipole- Permanent Dipole Attractions
- These usually occur in molecules which contain F, O, N and Cl. Aldhydes, Ketones, Esters and PVC are examples of these:
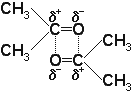
- Even though a bond in a molecule can be polar, the molecule itself, however, does not have to be polar.
- The shape of the molecule is an important factor in deciding whether a molecule is polar.
- Take for instance water:

- The oxygen atom has a greater electronegativity, thus there is a dipole in the O-H bond: hydrogen has a slight positive charge, and oxygen has a slight negative charge.
- One end of the water molecule is positive, and the other end is negative.
- However, if it wasn’t for the lone pair of electrons making the water into a v-shaped molecule, then the water molecule would be linear, therefore there would be no dipoles (both ends would be negative).
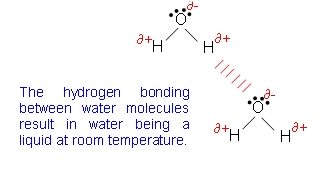
- This would make water a gas at room temperature.
- So, it is possible for a molecule to have polar bonds, and not be polar itself.
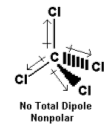
- Because the Cl atoms are distributed in a tetrahedral way around the carbon, the centre of the negative charge is halfway between all of the chlorines.
- It is at the centre of the molecule and therefore cancels the positive charge on the carbon. There is no overall dipole.
- Bond polarity depends on electronegativity differences; molecule polarity depends on electronegativity differences and the shape of the molecule.
Hydrogen Bonding
- Hydrogen Bonding is a strong intermolecular force that involves three features:
- A large dipole between a hydrogen atom and a highly electronegative atom.
- The small hydrogen atom which can get very close to other atoms.
- A lone pair of electrons on another O, N or F atom, with which the positively charged H atom can line up.
- In liquid HF, the hydrogen bonding is so strong, that the molecules form long chains.
- Dipole-dipole forces are present in all the hydrogen halides. The greater strength of the hydrogen bond can be seen when looking at their boiling points.
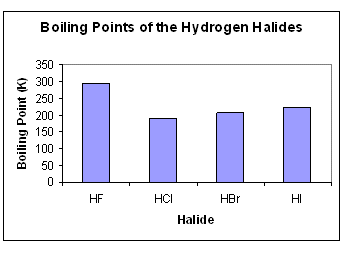
- Generally, as you move down the group, the molecules become bigger, meaning there are more electrons, so the instantaneous induced dipoles become stronger, hence the boiling point increases.
- However, HF has a higher boiling point than any of the other molecules; this is due to the strong hydrogen bonds between the molecules.
- Hydrogen bonding is also present in water:
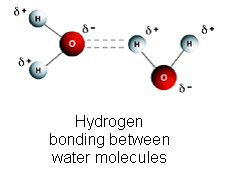
- Water molecules can form twice as many hydrogen bonds as hydrogen fluoride molecules. This is because the oxygen atom possesses two lone pairs of electrons with which the hydrogen atoms can interact.
- There are also twice as many hydrogen atoms than oxygen atoms- this maximises the bonding to the lone pairs.
- This makes water unique; in hydrogen fluoride there are three lone pairs of electrons, however there are only as many hydrogen atoms as there are fluorine atoms. This means that only a third of the available lone pairs can be used.
In NH3 there is only one lone pairs of electrons on the N, so on average only one of the three hydrogen atoms can form hydrogen bonds.
- In polymers, hydrogen bonds are responsible for the strong intermolecular forces between the polymer chains of many fibres.
- In nylon there are interactions between the H-atoms on the N-H groups, and the O atoms of the C=O groups.
Useful books for revision:
Revise AS Chemistry for Salters (Written by experienced examiners and teachers of Salter's chemistry)
Revise AS Chemistry for Salters (OCR) (Salters Advanced Chemistry)
Home