
Induced Polarisation
- Intermolecular forces are the forces between molecules.
- An induced dipole force arises because the electrons around atoms are in constant motion, and at a particular instant may not be evenly distributed.
- There are dipole forces between all atoms.
- Also, the electrons in one atom will repel the electrons in the neighbouring atoms, inducing a positive charge (the electrons are pushed away from the region).
- When this happens to an atom, we say that it has been polarised; an induced charge has arisen.
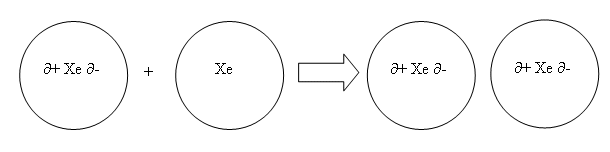
- The electrons in the xenon (left) have formed in a negatively charged electron cloud. The electron cloud on the negative end is dense (more electrons are in that area), whereas the electron cloud on the positive end is less dense (less electrons).
- The polarisation in the atoms is not permanent. Electrons are always moving around; the original polarity will be lost and different polarities will be formed.
- The intermolecular forces between xenon atoms are stronger than the intermolecular forces between helium.
- This is because xenon is larger than helium and it has more electrons.
- As it is larger, the electrons are not held as strongly by the positive nucleus, and so move more easily around the outside of the atom (forming electron clouds).
- The extra electrons mean that more dense electron clouds can form.
- Xenon atoms are more polarisable than helium atoms (can form bigger induced charges, therefore stronger intermolecular forces).
- Each time a xenon atom is polarised, there is a negative charge at one end, and a positive charge at the other; this is called a dipole.
- In xenon’s case it is an induced dipole, and so is temporary.
Molecular Shape has an Effect
- In structural isomers of branched alkanes, the atoms within the different isomers are identical; however in straight chains there is more contact between the atoms of different molecules.
- This means that there is more opportunity for an induced dipole force; straight alkane chains have stronger intermolecular forces between them than branched alkane chains; hence they have a higher boiling point.
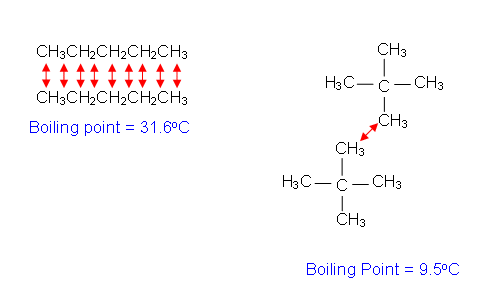
- Polymers have very long chain molecules; if the chains line up neatly, the intermolecular forces can be strong- making the polymer material strong.
Useful books for revision:
Revise AS Chemistry for Salters (Written by experienced examiners and teachers of Salter's chemistry)
Revise AS Chemistry for Salters (OCR) (Salters Advanced Chemistry)
Home



