
Hydrogen Bonding in Water
- Water has many unusual properties:
- Unexpectedly high values for the boiling point and enthalpy of vaporisation.
- A greater specific heat capacity than most other liquids.
- A decrease in density when it freezes.
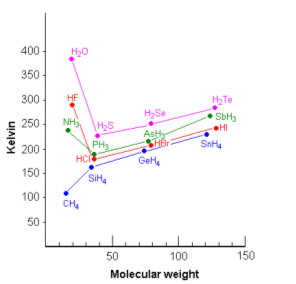
- The graph above shows how the boiling points of the hydrides changes as the molecular weight increases.
- The data from CH4 to SnH4 show the expected values for the boiling points; that is, as the molecular weight increases, the boiling point also increases.
- The points for H2O, HF, and NH3 are definitely out of line on the graph.
- This can be accounted to the hydrogen bonding.
- Hydrogen bonding holds molecules together stronger than any other type of molecular force. When the hydrides are boiled, the intermolecular forces holding them together must be overcome. As H2O, HF and NH3 have hydrogen bonds between the molecules; their boiling points are much higher than expected.
- The hydrogen bonding in H2O is particularly strong, this is because there are two lone pairs of electrons and two positive hydrogen atoms per oxygen atom. This maximises the intermolecular interactions.
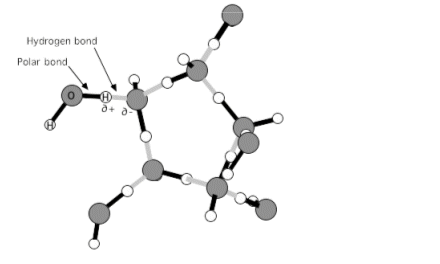
- The water molecules are close together in liquid form; however they become widely separated in a gas. For this to happen the intermolecular forces have to be overcome.
- The enthalpy of vaporisation is a measure of the energy needed to overcome these forces.
Specific Heat Capacity
- A measure of a substance’s ability to absorb heat energy (how many joules of energy are required to raise the temperature of 1 gram of a substance by 1K).
- Water has a very high heat capacity (4.18 joules g-1K-1).
- Water’s unusually high heat capacity can be related to hydrogen bonding; a lot of energy is required to weaken the hydrogen bonds within clusters of water molecules. This energy is not available for increasing the molecules kinetic energy (no available for raising the temperature); and so much of the energy is absorbed when breaking the hydrogen bonds.
Density of Water and Ice
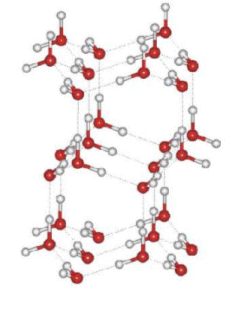
- When water is cooled to around 0oC it expands; this is an odd property that is not found in many other substances.
- This is because the hydrogen bonding gives ice an open structure with large spaces between some of the molecules.
- Therefore the density of ice is less than that of water, i.e. it floats!
- When the ice melts, the water molecules fall into some of the open spaces, thus giving it a greater density than what it came from.
Useful books for revision:
Revise AS Chemistry for Salters (Written by experienced examiners and teachers of Salter's chemistry)
Revise AS Chemistry for Salters (OCR) (Salters Advanced Chemistry)
Home





