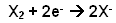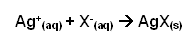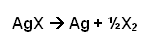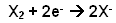Group 7- The Halogens
- The Halogens are found within the p-block in group 7. They all have 7 outer electrons, which is why they have similar properties. However, their masses increase as you move down the group, this explains their differences.
Properties of the Halogens

- The reactivity of the halogens decreases down the group (the size of the atom increases, therefore the positive charge is shielded more on the larger molecules, making it harder for it to gain an electron).
- Halogen does not dissolve in non polar substances, as they are themselves polar, however they do dissolve in water, and other polar substances.
Halide ions and silver ions
- The halide ions react with silver ions to form a silver precipitate. The colour of this precipitate becomes more substantial down the group. Chloride gives a white precipitate, Bromide a cream, and Iodide a yellow.
- The reaction with silver ions can be summarised with the ionic equation:
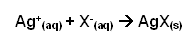
- The silver halide reacts with light going darker, which is why it is used for film in cameras.
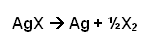
- The sensitivity to light decreases down the group.
Displacements Reactions
- The power of the oxidising agent decreases down the group, thus any halogen will displace a halide further down in the group.
- The table below shows the displacement reactions of potassium halides.
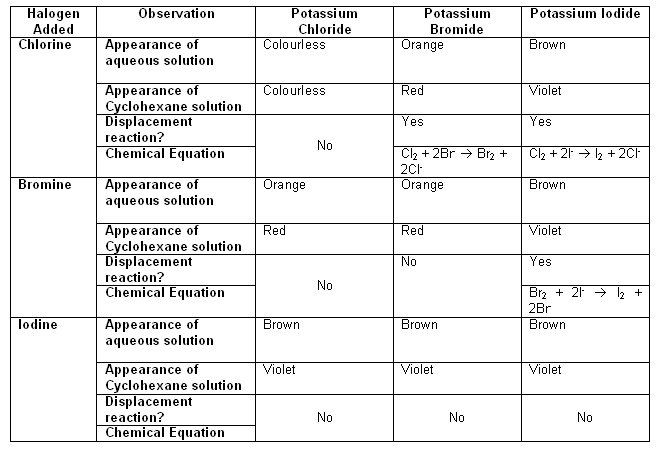
- Halogens working as oxidising agents: