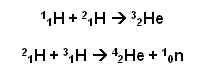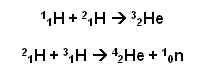Nuclear Fusion
- Fusion is an important type of reaction in which light nuclei are fused together to form heavier nuclei.
- High amounts of energy (millions of degrees Celsius) are required to overcome the positive charges on the two nuclei.
- Fusion is common within the stars, where heat energy is abundant.
Examples of Fusion reactions within the sun:
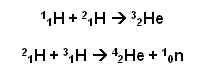
- The atomic numbers and mass numbers must balance within a nuclear reaction.
- When the universe was born, there was a lot of hydrogen.
- The hydrogen and dust are attracted to each other by gravity, forming huge areas of dust and gas called nebulas.
- The pressure in these nebulas causes extremely high temperatures.
- The high temperature allows the hydrogen nuclei to fuse with each forming helium.
- The nuclear fusion occurs fastest in the larger stars, as the temperature is greater.
- Most of the energy within stars is from the fusion of hydrogen to form helium; however there are other fusion reactions to form heavier elements.
- Small stars only convert hydrogen into helium.
- Medium sized stars like our sun (when hydrogen is depleted) convert helium into oxygen and carbon.
- Heavyweight stars convert helium into carbon and oxygen, followed by the fusion of carbon and oxygen into neon, sodium, magnesium, sulphur and silicon. Later reactions then convert these elements into calcium, iron, nickel, chromium, copper and others.
- In a heavyweight star; layers of elements are formed, with the denser elements closer to the centre. The element at the centre of the star is iron; this is because when iron nuclei fuse they don’t produce energy, but absorb energy. Eventually the centre of the star becomes unstable, and causes the star to explode. These explosions are called supernovae, the most violent events in the universe.
- After the explosions, the dust and gas is attracted together and the process of the star begins again.
Useful books for revision:
Revise AS Chemistry for Salters (Written by experienced examiners and teachers of Salter's chemistry)
Revise AS Chemistry for Salters (OCR) (Salters Advanced Chemistry)
Home