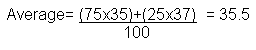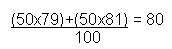Atomic Structure
| Relative Mass | Relative Charge |
| Proton | 1 | +1 |
| Neutron | 1 | 0 |
| Electron | 0 | -1 |
- Rutherford discovered the layout of the atom by firing alpha-particles at gold leaf particles.
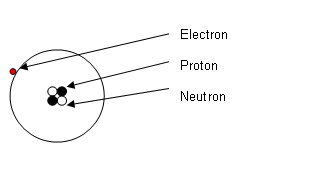
- The number of protons is a key number, it identifies every element. It is called the atomic number.
- Number of Protons + Number of Neutrons = Mass.
- Number of Protons = Number of Electrons.
- Relative Atomic Mass (Ar) = Average mass of an atom compared to 1/12 the mass of an atom of Carbon.
- Number of neutrons in an element can vary, which is why we have isotopes, for example there is chlorine-35 and chlorine-37.
- Chlorine 37 has an atomic mass of 37, and chlorine 35 has a mass of 35. The Relative Atomic mass of chlorine is said to be 35.5, because this is an average of the isotopes.
- 75% of chlorine atoms are Cl-35, and the other 25% are Cl-37
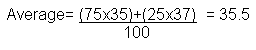
Working out the number of protons and neutrons examples:
| |
Protons |
Neutrons |
Electrons |
| 79Br |
35 |
44 |
35 |
| 81Br |
35 |
46 |
35 |
| 35Cl |
17 |
18 |
17 |
| 37Cl |
17 |
20 |
17 |
|
Bromine has two isotopes. 50% of bromine is 79Br, and the other 50% is 81Br. Work out the average mass:
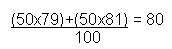
Useful books for revision:
Revise AS Chemistry for Salters (Written by experienced examiners and teachers of Salter's chemistry)
Revise AS Chemistry for Salters (OCR) (Salters Advanced Chemistry)
Home