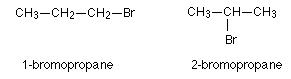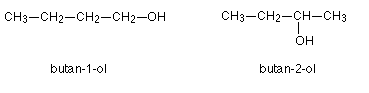Isomerism
- Two molecules which have the same chemical formula but differ in the way that their atoms are arranged are called isomers. Isomers are distinct compounds with different physical and chemical properties.
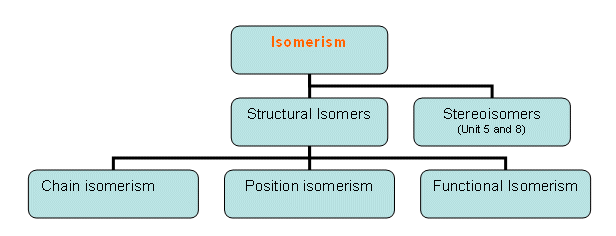
- Structural isomers have the same molecular formula, but have a different order in which the atoms are bonded.
Chain Isomerism
- These isomers occur because of the different possibilities of branching alkanes, for example there are two isomers of butane:
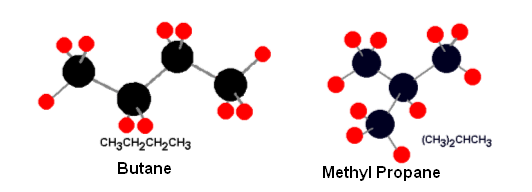
- Both these molecules have the same molecular formula, C4H10.
- Their different structures lead to different properties; Butane has a boiling point of 273k, and methyl propane has a boiling point of 261k.
- The octane number of a branched alkane is higher than that of a straight chain.
- As the number of carbons increases in the hydrocarbon, the number of isomers also increases.
Position Isomerism
- This is where an atom or group of atoms are replaced in the carbon chain or ring. These are called functional groups.
- The isomerism occurs when the functional group is situated in different positions in the molecule.
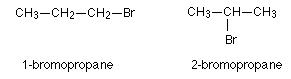
- 1-bromopropane, and 2-bromopropane each have the molecular formula C3H7Br; however the bromine atom is situated in different parts of the molecule.
- Another example occurs with alcohols:
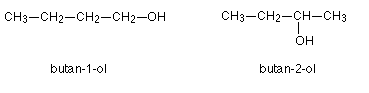
- Both have the same molecular formula C4H9OH, but the OH group is situated in different parts of the molecule.
Functional group isomerism
- It is possible for compounds with the same molecular formula to have different functional groups:
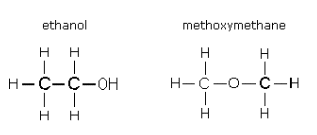
- C2H6O can either be an alcohol, or an ether depending upon its structural layout.
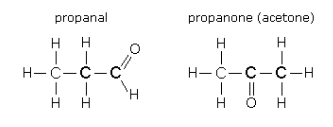
- The formula C3H6O can either be a ketone (propanal) or an acetone, depending upon the layout of the atoms within the molecule.
Useful books for revision:
Revise AS Chemistry for Salters (Written by experienced examiners and teachers of Salter's chemistry)
Revise AS Chemistry for Salters (OCR) (Salters Advanced Chemistry)
Home