
Entropy
- When petrol is spilt into an enclosed space such as a garage, the petrol vaporises and diffuses through the air to occupy all the available space.
- This is why petrol is a serious fire risk; it mixes with the air to form a highly flammable mixture.
- Chance and probability are the reason for diffusion.
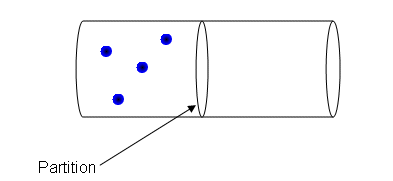
- In the diagram above, there is a container with a partition down the middle. The molecules in the left hand side of the container are constantly moving; travelling in a straight line until they hit another molecule or the side of the container.
- When the partition is removed, each of the molecules moving around can either end up in the left hand side of the container or the right.
- It is pure chance which side the molecules can end up in after a given amount of time.
- There are four molecules altogether and each one has two places they can be, so the total number of ways the molecules could arrange themselves is 24 = 16. Each of these ways is equally likely.
- Only one of these arrangements has all of the molecules in the left hand side of the container where they started. So the chance that the molecules will stay in one side of the container is 1/16.
- The molecules diffuse because there is more chance that they will spread out than stay in one place.
- In a real-life situation there will be billions upon billions of molecules instead of just five. So the possibilities of the arrangement of the gas are vast, meaning that there is an extremely small chance that the molecules will all stay in one place.
- The events that happen are the ones that are most likely to happen (the gas is more likely to spread throughout the container); the more ways an event can occur, the more likely it is to happen.
- The mixing of liquids is also another example of this rule. Liquids mix because there are more ways of being mixed than staying unmixed. In theory the liquids can stay unmixed, but the chance of this happening is extremely low.
- However, not all liquids mix; for example, petrol and water do not mix. This is because there is something to prevent the natural mixing process from happening.
- There are small forces between all of the molecules, but if the forces are stronger between one liquid than another, then the molecules will stay together, therefore not mixing.
- The general rule about mixing is that substances always tend to mix unless there is something stopping them (like attractive forces holding molecules together, preventing them from easily separating).
Measuring Entropy
- It is very important for chemists, biologists and physicists to be able to work out the number of ways an event can happen; so that they can work out how likely an event is to occur.
- Entropy is used to measure the number of ways an event can happen. The higher the entropy, the more number of ways there are for an event to happen.
- This means that generally the more disordered a system, the higher the entropy.
- Gases have higher entropies than liquids, which have higher entropy than solids.
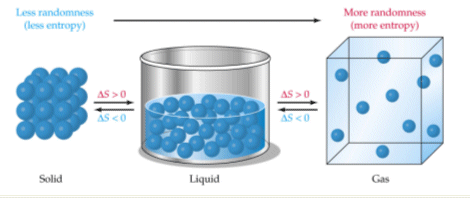
- This is because the molecules in gases are more randomly spread out than the water molecules which are more randomly spread out than the solid molecules.
- Substances with more complex molecules have higher entropy than substances with simple molecules.
Useful books for revision:
Revise AS Chemistry for Salters (Written by experienced examiners and teachers of Salter's chemistry)
Revise AS Chemistry for Salters (OCR) (Salters Advanced Chemistry)
Home



