
Rates of Reaction
Collision Theory
- The collision theory can be used to explain the factors affecting rates of reaction.
- The basic theory behind the collision theory is that chemicals react when their particles successfully collide.
- Take for instance the reaction between hydrogen and nitrogen to produce ammonia.
- The collision theory states that the reaction occurs when the hydrogen particles and nitrogen particles successfully collide.
- The more frequently they collide, the faster the rate of reaction.
Effect of Pressure
- The higher the pressure, the more particles there are per volume. If there are more particles in a space, then there will be more collisions within a given time period. This means that the rate of reaction will be higher.
Effect of Temperature
- When the temperature increases, the particles gain kinetic energy and move around faster. This results in more collisions.
- The speed of the molecules is proportional to the square root of the temperature (K).
- However, it’s not only how frequently the particles collide that matters, but also how much energy they collide with.
- The minimum energy needed for the reaction to occur is called the activation energy.
- Unless the particles collide with the energy equal to the activation energy, they just bounce off each other, and remain un-reacted.
- This is what happens most of the time during reactions, for example when N2 and H2 react at 300 K, only 1 in 1011 collisions results in a successful reaction.
- The collision theory states that reactions occur when molecules collide with a certain amount of energy. The more frequent these reactions, the faster the reaction.
Distribution of Energies
- At a temperature, the kinetic energy of the molecules varies; the different molecules move at different speeds.
- Some molecules have high kinetic energies, some have medium, and some have low kinetic energies.
- The Maxwell-Boltzmann distribution can be used to represent the distribution of kinetic energies.
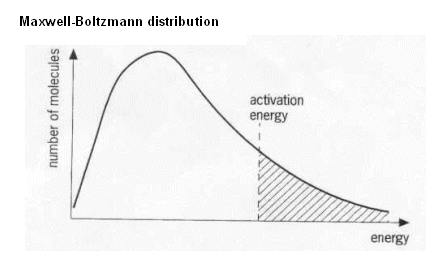
- As the temperature increases, the molecules kinetic energy increases; they move around faster.
- There is still a distribution of energies at higher temperatures, however a greater proportion of molecules have higher energies.
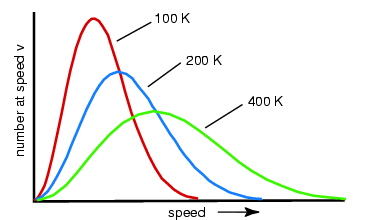
- These may look irrelevant to reaction rates; however they do have a purpose.
- If we add the activation energy to the distribution, then we can see how much of the distribution will result in successful collisions (below).
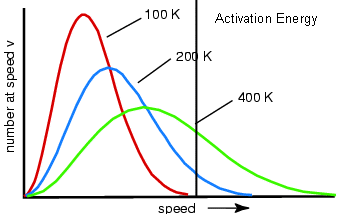
- The molecules to the right of the activation energy line can react, whereas the molecules to the left of the line are unable to.
- Notice that the distributions with higher temperatures have more of the graph to the right of the line; therefore more of the molecules react.
- This can be summarized by saying that reactions go faster at higher temperatures because a larger proportion of molecules have the required activation energy needed to react.
- Increasing the temperature may not make a difference to the energy of each individual molecule, but it makes a big difference to the proportion of the molecules.
Useful books for revision:
Revise AS Chemistry for Salters (Written by experienced examiners and teachers of Salter's chemistry)
Revise AS Chemistry for Salters (OCR) (Salters Advanced Chemistry)
Home






