
Identifying the Drug in Willow Bark
- Infrared Spectroscopy and Mass Spectrometry can be used to identify the chemical structure of compounds such as salicylic acid.
-
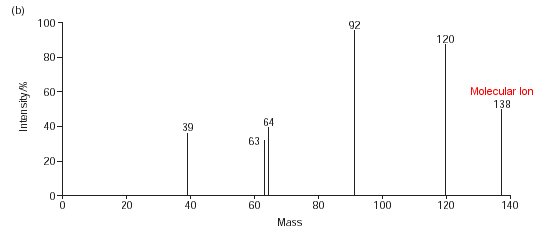
Below is the mass spectrum of salicylic acid; chemists can use this to find the structures of compounds.
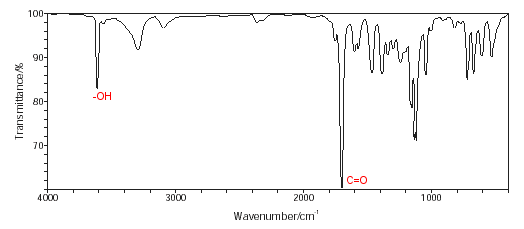
- The Infrared spectrum can also be used to provide important clues about the groups of atoms contained in salicylic acid.
Working out the structure
- Chemical tests show the presence of a phenolic OH group (test with FeCl3 and look for purple colour) and a COOH group (CO2 gas produced when mixed with carbonate).
-
Mass spectrometry shows that salicylic acid is the same compound as 2-hydroxybenzoic acid:
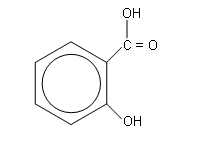
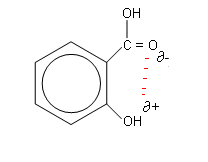
-
The mass spectrum fragmentation pattern shows that the structure cant be either of the two below, as a H2O has been lost from the spectrum, which cannot be lost from either of these molecules, as there must be hydrogen bonding occuring between the -COOH and -OH groups in order for a water fragment to be lost (above right):
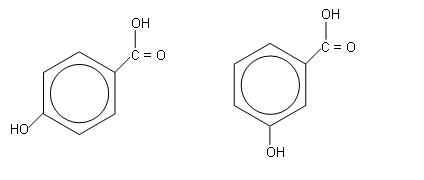
- Infrared spectroscopy shows the presence of an OH group and a C=O group.
Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)Salters (OCR) Revise A2 Chemistry