
Esters
- Esters are functional group isomers of carboxylic acids.
-
Esters are formed when a molecule of an organic acid reacts with an alcohol.
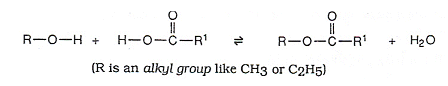
- This is an example of a condensation reaction as two smaller molecules have joined together to form a larger one, with the elimination of a small molecule (water above).
- A concentrated strong acid catalyst is usually present in the above reaction, as the esterfication process would be too slow without it.
-
Esters have strong, sweet smells. They are used widely in glues, as solvents, and in fragrances.

-
Esters are named from the alcohols and acids from which they are derived. For example,
Ethanol and Propanoic acid react to produce Ethyl Propanoate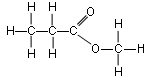
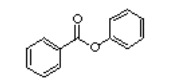
Esters from 2-hydroxybenzoic acid
-
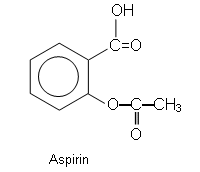
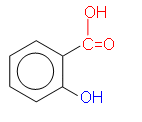 The structure of 2-hydroxybenzoic acid (right) makes it possible for there to be two ways in which it can be esterified. Either the OH group can be reacted with an organic acid to form an ester, or the COOH group can be reacted with an alcohol to form an ester.
The structure of 2-hydroxybenzoic acid (right) makes it possible for there to be two ways in which it can be esterified. Either the OH group can be reacted with an organic acid to form an ester, or the COOH group can be reacted with an alcohol to form an ester.
-
Aspirin is the product of reacting the –OH group with ethanic acid to form 2-ethanoylhydroxybenzoic acid.
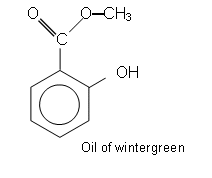
- The product of reacting the COOH group with methanol is methyl-2-hydroxybenzoate (oil of wintergreen).
-
Oil of wintergreen is used as a liniment; it is fat soluble and so can be absorbed through the skin. It also relieves pain and swelling like aspirin.
Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)Salters (OCR) Revise A2 Chemistry