
Genetic Engineering
- Scientists now know the genetic code for all the amino acids, and so if the order of amino acids within a protein is known, then it is possible for scientists to be able to make that protein using the genetic code.
- This is the basis of genetic engineering; genes are taken from cells of one type of organism that has a desired characteristic (e.g. insulin production) and are put into another type of organism.
- The details of the method vary according to the gene and micro-organism. Bacteria/yeast cells are usually the host cells, as they are easy to look after, their genes can be manipulated easily and they reproduce rapidly.
The general process
- The DNA for the desired gene is isolated in the donor cell (this can be isolated on the mRNA molecule or on a section of DNA).
- A restriction endonuclease is used to cut the gene from the DNA:
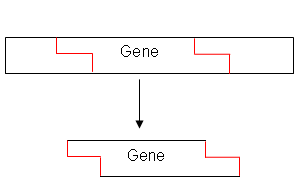 This leaves “sticky ends” on the gene (single strands of base sequences ready to hydrogen bond with complimentary bases.
This leaves “sticky ends” on the gene (single strands of base sequences ready to hydrogen bond with complimentary bases.
- The same restriction endonuclease is used to cut a section of DNA from a plasmid; this leaves complimentary base ends for the isolated gene to bond to.
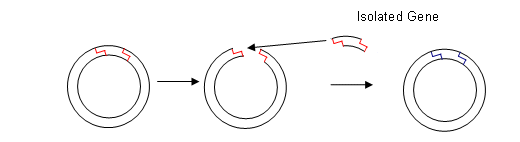
- The plasmids are then taken up by the host cells.

- The plasmid also contained a marker gene (this could have been a gene for antibiotic resistance, therefore the bacteria can be grown in antibiotics and any cell that hasn’t got the new plasmid will die).
- The bacteria cells multiply in a fermenter. If the new gene was for insulin production, the insulin would be produced within the fermenter and isolated later on.
What is genetic engineering being used for?
- Proteins such as insulin, human growth hormone and Factor 8 (blood clotting agent given to haemophiliacs) are products of genetic engineering.
- But the applications for recombinant DNA technology are far wider than just pharmaceuticals:
Vaccines
- The body’s immune system protects itself by recognizing proteins on the cell membrane of pathogens; the protein could be produced using genetic engineering, and a harmless version of the pathogen could be injected as a vaccine.
Waste management and pollution control
- Certain genetically modified bacteria can be used to convert harmful pollution into harmless waste. For example an oil digesting “super bacteria” has been formed from a collection of genes from different organisms, the resultant bacteria can consume all the chemicals within crude oil.
Materials
- The genes which code for the production of materials, such as spider silk or biodegradable plastic, can be isolated from organisms and added to bacterial cells, so that they can grow it, leading to easier extraction/greater production of the plastic.
Pest and weed control
- The gene for herbicide resistance can be placed in the wanted crop, this means that the crop wont die (but the weeds will) when a herbicide is applied.
- Genes for pesticide production can be added to plants, to save for the need to spray herbicides.
Selective plant breeding
- Genes for enhanced photosynthesis, or drought resistance can be added to plants to allow them to grow better in hostile regions. Nitrogen fixation genes can also be added, thus reducing need for artificial fertilizers.
Example of Genetic Engineering
- Insulin monomers work far better than insulin hexamers for diabetics, as they don’t have to be broken down and so work faster.
- Using genetic engineering, it has been made possible to alter the structure of the monomer, so that they don’t join together to form the hexamer.
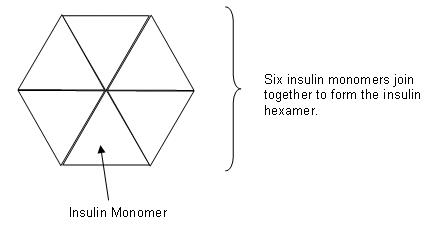
- The insulin monomers fit together so that the chemically identical faces are in contact.
- However, the monomers are chiral and so when they join together, the faces will be the opposite way around from each other; therefore the counterparts on each chain will not be next to each other.
- A closer look at the amino acid residues at the surfaces between the two monomers (dimerising surface), shows that most of them have non-polar side chains, the rest being polar or ionic.
- For example, residue B13 (amino acid 13 in the B chain) is a glutamic acid with a negatively charged side chain. This is placed next to residue B9, which is a serine with a polar side-chain.
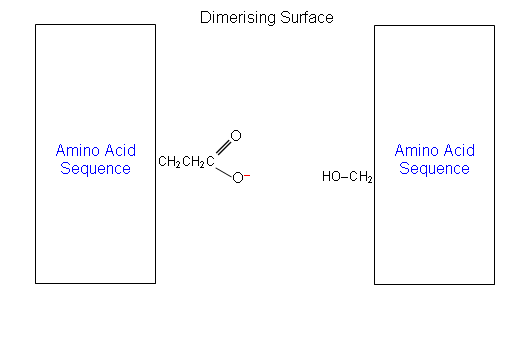
- If serine was replaced with aspartic acid, the four negatively charged COO- groups (two aspartic acid and two glutamic acid) would be placed together and the dimmer would be pushed apart:
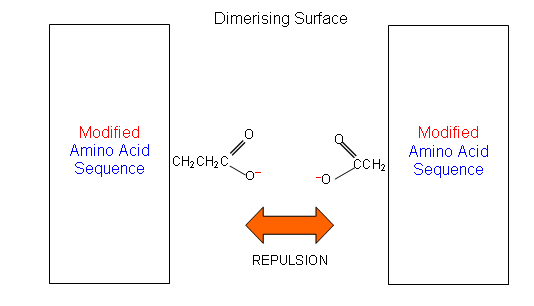
- Chemists can study the structure of proteins using computer programs. They can use the programs to determine how to change the structures of the protein and thus make them more desirable.
- The new form of insulin can be made by replacing the codon for serine with the codon for aspartic acid within the human insulin gene. We can then insert this gene into yeast/bacteria and they can make the adapted insulin for us.
- To do this, chemists first synthesise a section of the insulin gene which tells the cell to join together six amino acids of the B-chain around B9, exchanging the codon for serine with that for aspartic acid:
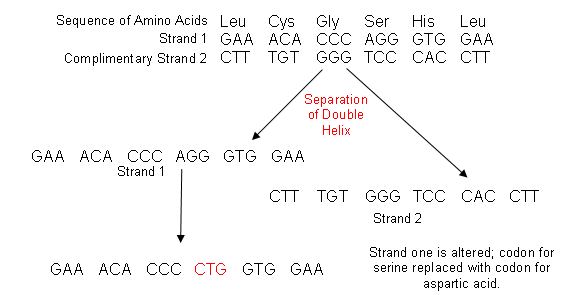
- Strand two then joins up with the modified strand one:
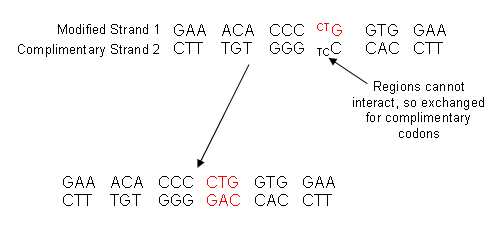
Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)
Salters (OCR) Revise A2 Chemistry Home
Home











