Enzymes
- Enzymes are biological catalysts that speed up chemical reactions. Enzymes speed up chemical reactions in the body by a factor of between 106 to 1012 times, allowing vital chemical reactions to take place at normal temperatures
- There are around 40,000 enzymes in the human body.
- Enzymes are complex structures of three-dimensional proteins (quaternary protein structure). They are many times larger than the molecules involved in the reactions they catalyse.
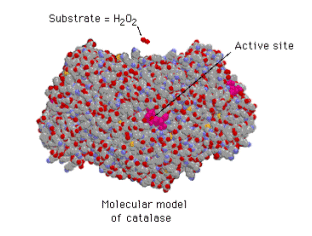
- The reaction takes place in a small area of the enzyme called the Active Site, the rest of the protein acts as scaffolding holding the protein together.
- The arrangement of the amino acids in the active site makes it specific for only one type of substrate. The enzyme is complementary in shape, charge and hydrophobic/hydrophilic areas.
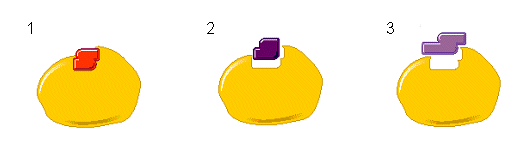
- Even when other substrates are present (2 and 3) only the substrate with the specific shape will bind to the enzyme (1). The enzyme and substrate are like jigsaw pieces that fit together.
- When the substrate and enzyme “collide”, hydrogen bonds or interactions between ionic side groups hold the substrate and enzyme together. After the new products are formed, they are released and the enzyme’s active site is free for more substrates to bind to.
Changing the pH
- Enzymes are very sensitive to any changes in pH. This is because the change in concentration of H+ ions affects the boding holding the tertiary structure together.
Changing the temperature
- Enzymes are denatured at higher temperatures. This is because high temperatures break the hydrogen bonding holding the tertiary structure together.
Uses of Enzymes
- As enzymes are so specific, they are very good at detecting certain reagents; for example a diabetes test strip can use an enzyme to detect the presence of glucose.
- There is a wide range of medical diagnostic kits based upon the use of enzymes.
- However, the medical industry only uses small quantities of enzymes.
- Most enzymes are used in the food industry and in the manufacture of washing powder.
- Most of the enzymes used in these industries are hydrolases (enzymes which hydrolyse fat, proteins or carbohydrates).
- In the food industry, enzymes are mainly used for:
- Producing glucose syrup by breaking down starch.
- Making cheeses using rennet enzymes which break down casein (milk protein) and cause the separation of the curds from the whey.
- Baking, brewing and fruit processing.
- Enzymes in washing powder include:
- Protease- to break down the protein in blood stains.
- Lipase- to break down fats.
- Genetic engineering is being used to make thermostable proteins that can be used in hot washes.
Enzymes as catalysts
- A catalyst is a substance that alters the rate of a reaction without being used up itself in the process.
- Most catalysts speed up the rates of reactions.
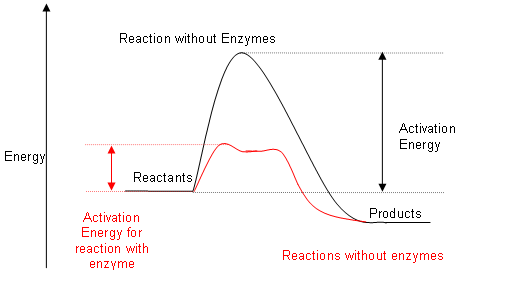
- They do this by providing an alternate reaction pathway that has a lower activation energy.
- Enzymes are very useful as catalysts; without them reactions that occur within our body would need to take place at extremely high temperatures, rather than just 37oC.
The energy profile diagram below shows the effect of enzymes on the activation energy of a reaction:
- Enzymes are usually only present in small traces. If the substrate concentration is high enough, then all of the enzymes will have a substrate attached to them (forming an enzyme substrate complex).
- If the substrate concentration is increased anymore, then no further enzyme substrate complexes can be formed, thus the rate of reaction does not increase.
- In this situation, the rate of reaction does not depend on the substrate concentration, which is thus zero order with respect to the substrate concentration.
- When the substrate concentration is low enough, not all of the enzyme active sites are being used, thus an increase in substrate concentration will result in an increase in the rate of reaction.
- In this situation, the rate of reaction depends directly on the substrate concentration, hence it is a first order reaction with respect to substrate concentration.
Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)
Salters (OCR) Revise A2 Chemistry Home
Home