


 Salt + Water
Salt + Water
For Example,
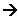 CH3COO-Na+(aq) + H2O(l)
CH3COO-Na+(aq) + H2O(l)
 Salt + Carbon dioxide + Water
Salt + Carbon dioxide + WaterFor Example,
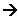 CH3CH2COO-Na+(aq) + H2O(l) + CO2(g)
CH3CH2COO-Na+(aq) + H2O(l) + CO2(g)
 Metal Salt + Hydrogen
Metal Salt + Hydrogen
For Example,
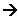 (COOH)2Mg(aq) + H2(g)
(COOH)2Mg(aq) + H2(g)
 Ester + Water
Ester + Water
For Example,
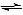 CH3COOCH2CH3 (aq) + H2O(l)
CH3COOCH2CH3 (aq) + H2O(l)
For Example,
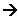 CH3NH3+(aq)
CH3NH3+(aq)
For Example,
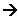 CH3CONHCH2CH3 (s) + HCl(aq)
CH3CONHCH2CH3 (s) + HCl(aq)
For Example,
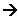 CH3COO-Na+(aq) + C6H5NH2(aq)
CH3COO-Na+(aq) + C6H5NH2(aq) For Example,
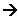 CH3COOH(aq) + C6H5NH3+Cl-(aq)
CH3COOH(aq) + C6H5NH3+Cl-(aq)