
Stereo Isomerism
- There are two main types of isomerism, structural isomerism and stereo isomerism.
- Structural isomerism is concerned with the order in which the atoms are bonded and is made up of chain isomerism, position isomerism and functional group isomerism.
- Stereo isomerism is concerned with the layout of the atoms in space; it is made up of geometric isomerism (e.g. cis-trans), and optical isomerism.
Geometric Isomerism
- But-2-ene is a structural isomer of but-1-ene.
- However, if you look at how the but-2-ene can be formed, then you will realize that there are two variations of the molecule:

- To turn the first molecule into the second; on end of the molecule must be rotated; however this would involve breaking one of the bonds in the C=C double bond.
- This would require a chemical reaction, and so the two variations of but-2-ene are isomers.
- They are cis and trans isomers.
Cis and Trans Isomers
- Cis and trans isomers are molecules with the same atoms within them, but a different layout of the atoms. The order of the atoms in cis trans isomers is the same, but the geometries are different.
- This form of isomerism is sometimes called geometric isomerism, and is a type of stereo-isomerism.
- Stereo-isomers have the same molecular formula, and the atoms are bonded together in the same order; however the arrangement of the atoms in space is different in each isomer.
- Cis and trans isomers are given different names- if the substituent groups are on adjacent sides of the double bond to each other, the prefix cis is added. If they are on opposite sides, the suffix trans is added.
- They must have different groups on each C of the double bond.
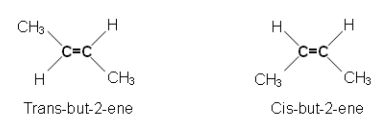
- Cis and Trans isomers can also be represented by skeletal formula.

- As cis and trans isomers are different compounds, they have different physical properties.
Optical Isomerism
- When four single bonds are formed around a central carbon atom, the molecule formed is tetrahedral in shape.
- If the four groups are all different, the molecule can exist in two isomeric forms. This can be seen with the amino acid, alanine:
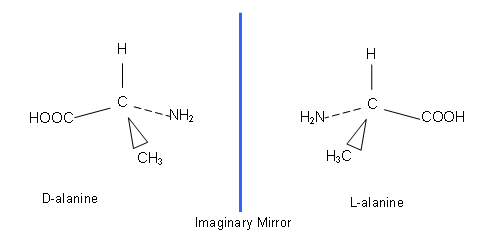
- The way that they are different is that they are mirror images of each other which can’t be superimposed onto each other.
- In the two molecules, the H atom, the C atom and the CH3 groups will coincide (in the same planes as each other); however, the COOH and NH2 groups are in the wrong places; no amount of twisting will allow them to be superimposed onto each other.
- The only way to make them the same is to break the C-COOH and N-NH2 bonds and swap the groups over; this involves bond breaking and bond making and so is a chemical reaction.
- All molecules have mirror images; however some of them don’t exist as non super-imposable images:
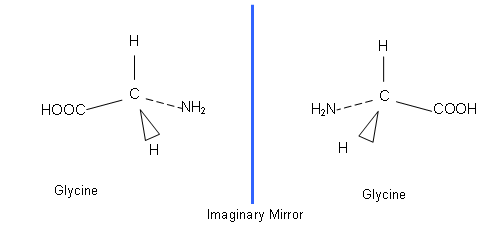
- The mirror images of glycine can be superimposed onto each other; the molecule just has to be flipped vertically and the isomer is made.
D/L enantiomers
- Molecules that show this kind of isomerism are called enantiomers.
- Any central carbon atom that is surrounded by four different atoms/groups will form two enantiomers.
- The arrangement of atoms like this is described as a chiral centre (our hands and feet are also examples of chiral centres, which is where the word chiral comes from- Greek for “handedness”).
- As many proteins have four different groups around a central carbon atom, they also show chirality.
- All the proteins within our body are built up from L-enantiomers (they have the same geometric arrangement around the central carbon atom).
- L-enantiomers can be identified using the CORN rule: with the H atom pointing upwards, looking down from the H atom to the carbon and moving clockwise, the L-amino acid has the order COOH, R, NH2, i.e.
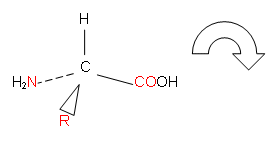
- Enantiomers behave identically in all chemical reactions and most of their physical properties are the same.
- The only difference comes when they are in the presence of other chiral molecules.
- D and L enantiomers will react differently with our enzymes (like trying to put a left hand glove onto your right hand).
- Examples of different effects include:
- Enantiomers react differently with chiral taste-buds (D amino acids are sweet and L-amino acids are bitter/tasteless).
- Enantiomers can smell differently.
- Many enantiomers are beneficial medicines; however their isomers can have drastic effects (e.g. thalidomide).
- D-amino acids do exist in nature, for example penicillin works by breaking down the peptide links in D-alanine; these occur in the cell walls of bacteria but not in humans. Thus penicillin only affects bacterial cells.
Test for enantiomers
-
A plane of polarised light is rotated in opposite directions by each enantiomer.
-
If a solution containing equal amounts of each enantiomer is placed in a polarimeter, the solution will be optically inactive.
Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)
Salters (OCR) Revise A2 Chemistry Home
Home







