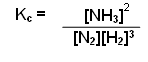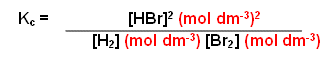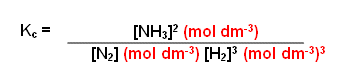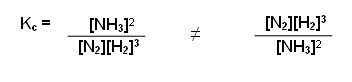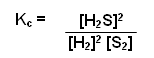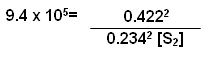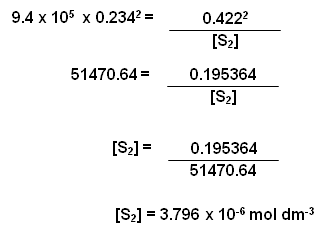H2(g) + I2(g)
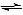 2HI(g)
2HI(g)
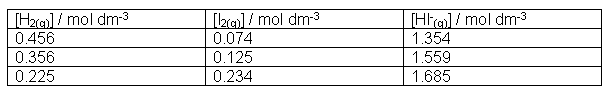
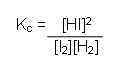
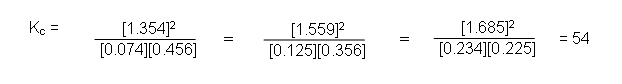
aA + bB
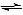 cC + dD
cC + dD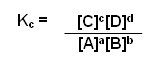
- Write a balanced equation for the reaction.
- Take the concentrations of the products and raise them to the power of the number which appears in front of it in the balanced equation.
- Divide them by the concentration of reactants raised to the power of the number in front of them in the balanced equation.
For example:
Write down an equation to show the equilibrium constant for the formation of ammonia from nitrogen and hydrogen:
- Write a balanced equation:
N2(g) + 3H2(g)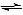 2NH3(g)
2NH3(g)
- Raise the concentration of the products to the power of the number in front of them:
[NH3]2
- Divide them by the concentration of the reactants raised to the power of the number in front of them: