
Partition Equilibria
- In addition to phase equilibria (between gaseous and liquid state), and chemical equilibria, there is also an equilibrium that exists between solutes in different solvents.
- Iodine is a Group VII liquid that dissolves in both inorganic and organic solvents, such as potassium iodide and tetrachloromethane.
- When iodine is dissolved in potassium iodide, the solution is brown in colour:

- If this solution is then added to a separating funnel with CCl4, the iodine passes between the two liquids until an equilibrium is established:
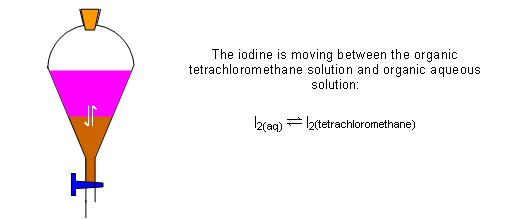
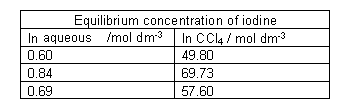
- The layers could be run into separate containers and the concentration of iodine in each solvent determined:
- This can be written as an equilibrium constant:
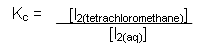
- The value for the equilibrium constant comes out at around 84. This particular kind of equilibrium constant measures how a substance distributes (partitions) itself between two solvents. It is called a partition coefficient Kow.
- The partition coefficient in the above example is determined by the fact that iodine is a non-polar substance and therefore prefers to be dissolved in an organic substance.
Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)
Salters (OCR) Revise A2 Chemistry Home
Home








