
Bonding, Structure and Properties
- The bonding and structure of substances determine their properties.
- Bonding is the way the atoms are held together.
- The structure is the way the atoms are arranged relative to each other.
- There are two major types of structure: giant and molecular.
- Giant structures go on indefinitely, whereas molecular structures are made up of groups of atoms.
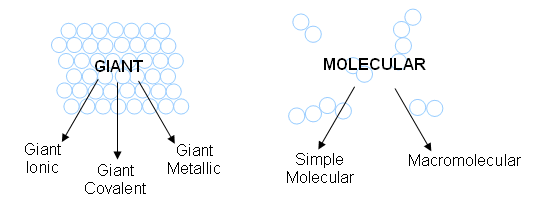
- Three main factors are important in deciding the properties of a substance:
- The type of basic particles it contains. The substance could contain atoms, ions or molecules. For example, if it contains ions (such as sodium chloride), then it will conduct electricity when molten or dissolved in water. To be soluble in water, the substance must contain either ions or polar molecules.
- The way the basic particles are bonded together. The bonding may be ionic, covalent, metallic or weak intermolecular forces. The stronger the bonds, the higher the melting/boiling point and the hardness of the substance.
For example, silica, SiO2, has strong covalent bonds linking every atom to several others forming a giant covalent structure. The atoms in silica are very hard to separate, and therefore it is very hard and difficult to melt. Carbon dioxide on the other hand has strong covalent bonds between the C and O atoms, but only weak intermolecular forces between each CO2 molecule. The molecules are therefore easily separated and so CO2 has a low melting/boiling point.
- The way the particles are arranged relative to one another. The particles may be arranged in 1-dimensional plains (such as in polymers), 2-dimensional sheets (like clays) or in many different kind of 3-dimensional arrangements.
Graphite is arranged in 2-dimesnional sheets, and so its layers can move past each other (think of writing using a graphite pencil). Diamond, on the other hand, has a giant 3-dimensional structure and is the hardest natural substance.
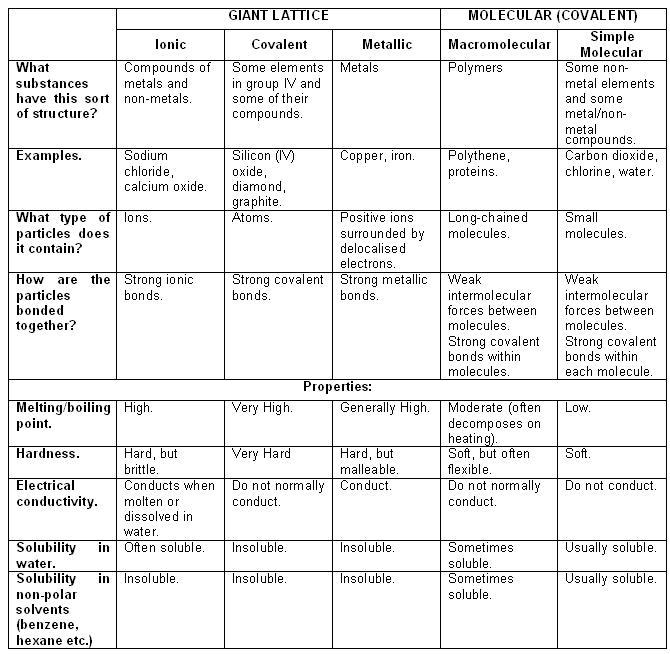
- The tables below summarises the different structures and their main properties:
Structure and the periodic table
- There is a trend in the structure of the elements as you move from left to right across a period of the periodic table.
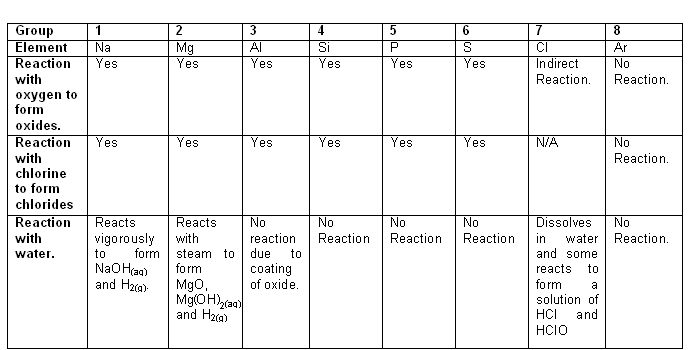
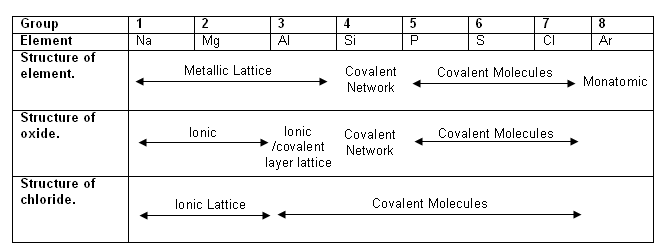
- The trends in the structures of the oxides and chlorides are closely linked to the trends in their properties; for example the acid-base character of the oxides and the behaviour of the chlorides in water.
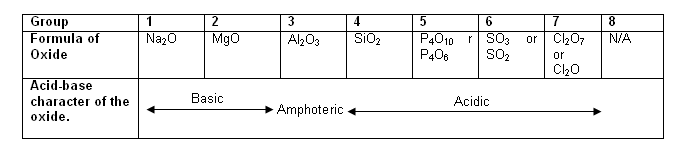
Acid-base properties of oxides
- It is evident from the following table, that the acid-base properties of the oxides are linked with the structures of the oxides (previous table).
- The general pattern is that oxides with giant ionic lattices are basic, whereas structures with covalent structures are acidic.
- The bonding in Al2O3 is both ionic and covalent in nature; aluminium oxide is thus amphoteric.
Behaviour of the chlorides in water
- The behaviour of the chlorides with water is also closely linked to their structure. The general pattern is for chlorides with giant ionic lattices to simply dissolve in water (no chemical reaction), whereas the chlorides with covalent molecular structures react, producing fumes of hydrogen chloride and forming acidic solutions.
- The covalent chlorides are hydrolysed in water:
AlCl3(s) + 3H2O(l) 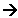 Al(OH)3(aq) + 3HCl(g)
Al(OH)3(aq) + 3HCl(g)
Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)
Salters (OCR) Revise A2 Chemistry Home
Home






 Al(OH)3(aq) + 3HCl(g)
Al(OH)3(aq) + 3HCl(g)
