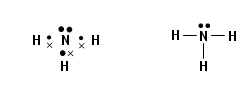Group V and Nitrogen
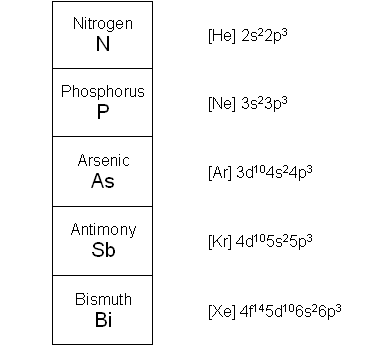
- Group V of the periodic table shares similar characteristics of the other groups of the p-block; at the top of the groups are the non-metals (nitrogen and phosphorus) and at the bottom of the group are the metalloids (antimony and bismuth).
- Atoms of the Group V elements can form three covalent bonds by sharing the three unpaired p-electrons.
- This gives them compounds in which the Group V element has an oxidation state of +3 or -3.
- The lone pair of electrons that each element has allows them to form dative bonds. This enables them to form compounds in which their oxidation state is five.
- Nitrogen and phosphorous are very important elements within group five; they are constituent elements in living things; they are both essential for healthy plant growth.
Nitrogen
-
Nitrogen is the most abundant gas in our atmosphere; however it is extremely unreactive.
- This low reactivity arises due to the strong triple bond between nitrogen atoms in N2 molecules.

- In order for nitrogen to react, the triple bond between the nitrogen atoms must be broken, or partially broken.
- This requires large amounts of energy, thus the activation energy for reactions involving nitrogen is very high.
- During thunderstorms, the highly energetic lightening flash can provide enough energy to form nitrogen oxides from atmospheric oxygen and nitrogen.
- Once reacted, it can form many useful compounds, for example ammonia, nitrogen oxides and nitrates.
Ammonia
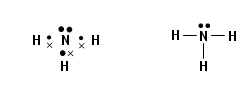
- Ammonia is an example of a nitrogen hydride.
- The lone pair of electrons on the ammonia allows it to acts as a nucleophile (donating a pair of electrons to a positively charged carbon atom), as a base (forming a dative bond with an H+ ion) and as a ligand.
Nitrogen Oxides
- Nitrogen forms many different oxides, all of which are gaseous.
- Some of the more important ones are:
-
Nitrogen monoxide (NO), a colourless gas which turns to brown NO2 in air. It is produced in combustion processes, especially from internal combustion engines. It is also formed in thunderstorms, and in the soil by denitrifying bacteria.
- Nitrogen dioxide (NO2) is a brown gas; it is toxic and is also formed in combustion processes and thunderstorms.
- Dinitrogen oxide (N2O) is a colourless gas formed in the soil by denitrifying bacteria.
Nitrates
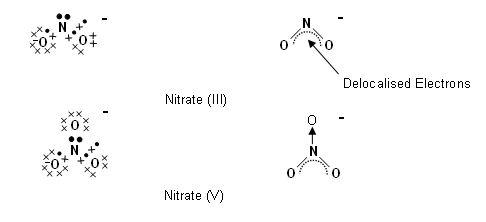
- There are two types of nitrate ions in the nitrogen cycle: nitrate (III), NO2-, and nitrate (V), NO3-.
- Their names are distinguishable from one another by their oxidation states.

Home