
Ions in Solution
- There are two important factors in deciding how ions behave in solution and how they behave in ion-exchange; the size of the ion and the charge on the ion.
- For example, if an ion is highly charged, it will be strongly attracted to oppositely charged ions in the solution and polar molecules (water for example).
- If the ion is small, it will be more strongly attracted to the oppositely charged ions and polar molecules, as it will be able to get in close to them.
- If an ion is both highly charged and small, it can be classed as having a high charge density.
- Ions with a high charge density tend to:
- Attract water molecules strongly and become very hydrated.
- Attract oppositely charged ions strongly, forming ionic lattices with large lattice energies and therefore high boiling and melting points.
Size of atoms and ions
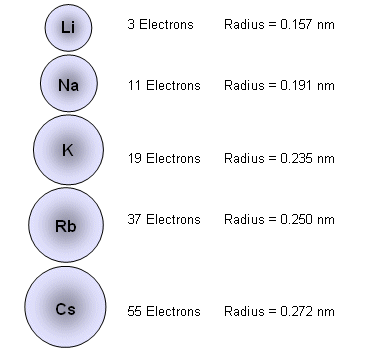
- The electrons take up the majority of the space in atoms and ions; therefore the more electrons there are in an ion or atom, the larger it will be.
- For example, in Group I, the atomic radii of the atoms increases down the group as more electrons are added:
- Moving across a period, the atomic radii decreases even though extra electrons are being added.
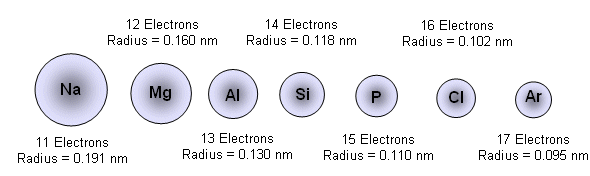
- This is because the number of protons increases along the period.
- As more protons are added, the nuclear pull on the electrons increases; as the electrons are being added to the same shell across each period, the atomic radii decreases as the extra charge pulls the electrons closer to the nucleus.
- For example, in period three:
- At potassium, the atomic radii is much larger; this is because a new electron shell is started- the fourth shell.
- The fourth shell is further away from the nucleus and so potassium has a larger atomic radius.
Ions
- Atoms become ions when electrons are added or taken away; this affects the size of the atomic radius.
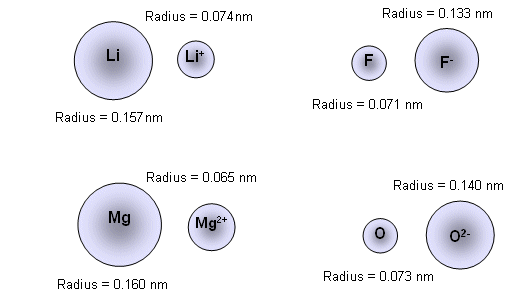
- Adding an electron to an atom (producing a negative charge) results in the ion being larger than the atom. Adding two electrons has an even greater effect.
- Removing an electron results in an ion that is smaller than the atom. Removing more than one electron has an even greater effect.
- The general rule is:
r2+ < r+ < r(atom) < r- < r2-
- This effect of charge can be illustrated in the following examples:
Hydration
- The above situation works for ions in the solid, liquid and gaseous phases; however, when an ionic substance is dissolved in water, the situation is different.
- In water, the ions attract water molecules and become hydrated; this makes the ion bigger, as there will be a layer of water molecules around it.
- The higher the charge density of the ion, the more water molecules it can attract, and therefore the bigger it becomes in water.
- So, ions which are small in the absence of water become large when in the aqueous phase.
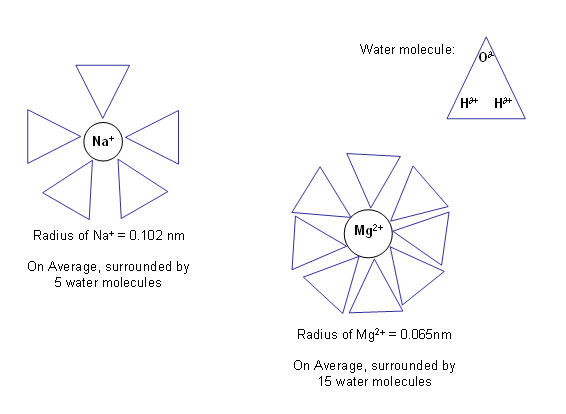
- Hydration makes small, strongly attractive ions behave as if they are larger and less attracting.
Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)
Salters (OCR) Revise A2 Chemistry Home
Home






