
Solubility Products
- Nothing is insoluble; there will always be some substance dissolved in solution, no matter how small.
- The increase in entropy associated with dissolving ensures that this is so.
- Thus, even on a beach, rain water will leach out the silicon dioxide and transport it to the sea.
- A sparingly soluble ionic solid, such as calcium carbonate, in contact with a saturated solution of its ions is an example of the following equilibrium:
CaCO3(s) + aq 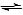 Ca2+(aq) + CO32-(aq)
Ca2+(aq) + CO32-(aq)
- As the solution is saturated, adding more solid will not push the position of equilibrium any further to the right; therefore the solid can be neglected from the expression for the equilibrium constant; this gives the following equilibrium constant:
Ksp = [Ca2+(aq)] [CO32-(aq)]
- This is known as the solubility product.
- There are solubility product values for all sparingly soluble solids, for example:

- We can use the Ksp values to calculate the concentration of a particular ion within a saturated solution; for example, the concentration of chloride ions in a saturated solution of AgCl:

- Equal amounts of Ag+ and Cl- must go into the solution, so:

- The Ksp value can also be used to determine the how other ions in the solution can affect the solubility of a sparingly soluble compound, for example, how adding extra Cl- or Ag+ ions will effect the concentrations.
- Imagine that we added some silver chloride to a 0.1 mol dm-3 solution of sodium chloride; the concentration of dissolved silver ions at equilibrium can be worked out.
- We have to assume that the all of the Cl- ions in the solution come from the sodium chloride (this can be done, as only a small amount of Cl- ions in the solution will come from the silver chloride). So therefore, the concentration of Cl- ions is 0.1 mol dm-3:

Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)
Salters (OCR) Revise A2 Chemistry Home
Home
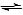 Ca2+(aq) + CO32-(aq)
Ca2+(aq) + CO32-(aq)





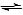 Ca2+(aq) + CO32-(aq)
Ca2+(aq) + CO32-(aq)



