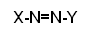Synthetic Dyes
Alizarin
- Mauve was the first synthetic dye ever produced; it was synthesised by William Perkin in 1856.
- Up until Perkin had produced the first synthetic dye, plants were the main source for colouring material.
- For example, the indigo plant was used to make blue dyes and the madder root was used to make red dyes.
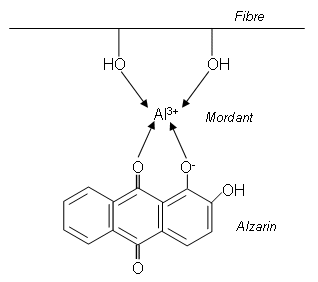
- The colouring matter from the madder root is a substance known as alizarin; alizarin only sticks to material that has been impregnated with a metal compound such as aluminium sulphate.
- This process is known as Mordanting, and the colour of the material depends upon the metal used; with an aluminium mordant the dye is red, with tin(ii) the cloth is pink, and iron(ii) gives a brown colour.
- Mordanting takes place under alkaline conditions so that a metal hydroxide is precipitated in the fibres; the metal ions firmly attach themselves to the cloth and then bind to the dye molecules by forming chelate rings (complex where ligands are datively bonded to a central metal ion):
- Even though alizarin had been used for thousands of years, its structure was not discovered until 1868, when Carl Graebe and Adolf Bayer proved that alizarin was derived from anthracene, a minor component of coal tar.
- From 1868-69, Graebe and Bayer developed a method for the production of alizarin.
- In 1868, a crop of 70,000 tonnes of madder root was processed to produce around 700 tonnes of alizarin; by 1873, the madder fields had disappeared.
Azo Dyes
- The first azo dyes were synthesised by coupling a diazonium salt (obtained from phenyl alanine) with one of a variety of coupling agents.
- Otto Witt, a Swiss trained chemist, completed research into why the aromatic azo compounds were coloured.
- He put forward the theory that a dye molecule is built up from a group of atoms called a chromophore, which is responsible for its colour.
- It is now known that chromophores usually contain unsaturated groups, such as C=O, -N=N- which are often part of an extended delocalised electron system involving arene ring systems.
- For example, Chrysoidine, an orange dye, is made up of a chromophore with a delocalised system:
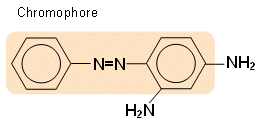
- Attached to the chromophore in chyrsoidine are the two amine groups; these interact with the chromophore to produce the orange colour.
- Other functional groups can be added to:
- Modify/enhance the colour of the dye.
- Make the dye more soluble in water.
- Attach the dye molecules to the fibres of the cloth.
- All azo dyes have the same common structure:
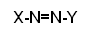
- Chemists worked to produce as many XY combinations as possible to make a wide range of new dyes with good colours, which would fast to fabrics and were commercially viable.
- A vast range of azo dyes are now available by coupling one of fifty diazonium salts with one of fifty two coupling agents. Most of the colours formed are yellow, orange or red.
Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)
Salters (OCR) Revise A2 Chemistry Home
Home