
The Chemistry of Colour
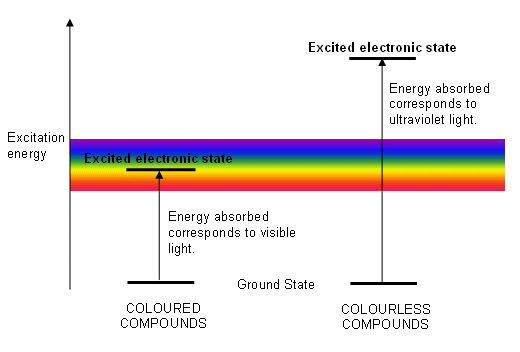
- Substances are coloured if they absorb energy that is in the visible frequency of the electromagnetic spectrum.
- The energy absorb causes electronic changes; electrons are promoted from their ground state to a higher energy level (they become excited).
- The difference in energy between the ground state and the excited state is equal to the quantum of energy absorbed and so determines the wavelength of light absorbed.
- It is the outermost electrons that are excited; these are the electrons involved in bonding or in lone pairs.
- The inner electrons are held tightly by the positively charged nucleus; the energy needed to promote these is very large.
- Not all electronic transitions are brought about by visible light; some transitions require more energy and are brought about by absorbing light in the ultraviolet region of the spectrum.
- If only light from the ultraviolet region of the spectrum was absorbed, the compound would appear colourless.
- The energy needed to excite an electron in a coloured compound is referred to as the excitation energy:
Coloured Organic Compounds
- These often contain unsaturated groups such as C=C, C=O or -N=N-.
- These can form part of an extended delocalised electron system called a chromophore.
- Electrons in double bonds are more spread out than those in single bonds, and therefore require less energy to become excited; this means that the energy absorbed is within the visible region rather than in the ultraviolet region of the electromagnetic spectrum.
- Functional groups, such as –O-H, –NH2, or –NR2 are often attached to the chromophores to modify the colour of the molecule; these groups all contain lone pairs of electrons which become involved in the delocalised electron system.
- Small changes to the delocalised system can change the energy of light absorbed by the molecule, thus changing its colour.
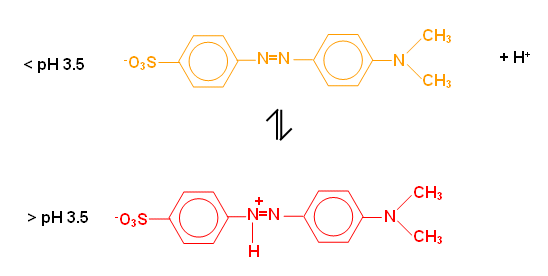
- Dye molecules, such as methyl orange, can have a different colour in acidic or alkaline conditions; compounds like this are often used as acid-base indicators:
Coloured inorganic compounds
- Transition metals are often present in coloured in organic compounds.
- Ligands datively bonded to the transition metal cause the splitting of transition metal’s d-sub shell into two energy levels.
- The d-orbitals are partially filled.
- Electrons are promoted from the lower energy level to the higher one, absorbing the frequency of light corresponding to the difference in energies between the two energy levels.
- The frequency of light absorbed corresponds to visible region of electromagnetic spectrum.
- Therefore this light is absorbed, so solution appears coloured.
- Sometimes the absorption of visible light can cause the transfer of an electron from the ground state of one atom, to the excited state of an adjacent atoms; this is referred to as electron transfer.
- Electron transfer is responsible for many bright pigments, such as Chrome Yellow and Prussian Blue.
Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)
Salters (OCR) Revise A2 Chemistry Home
Home



