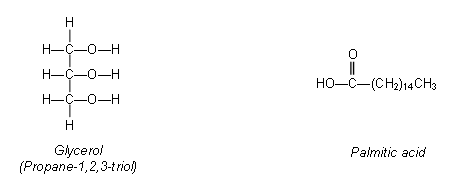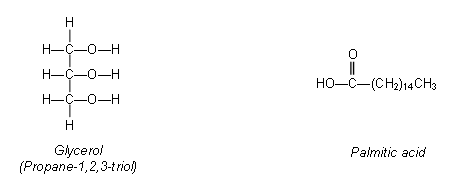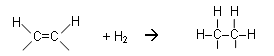Oils and Fats
- Oils/fats are important molecules for storing chemical energy in living systems.
- Oils and fats have the same basic structure; however fats are solid and oils are liquid at room temperature.
- Oils and fats are esters of glycerol (propane-1,2,3-triol) with long chained carboxylic acids, for example:
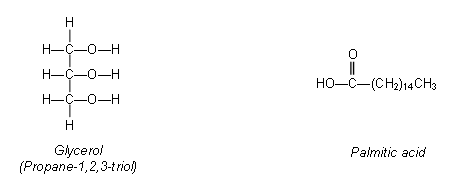
- Glycerol can react with three palmitic acid molecules to form a triester:
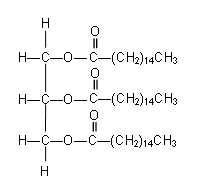
- The triesters found in natural oils are usually mixed triesters (the three fatty acid groups are different:
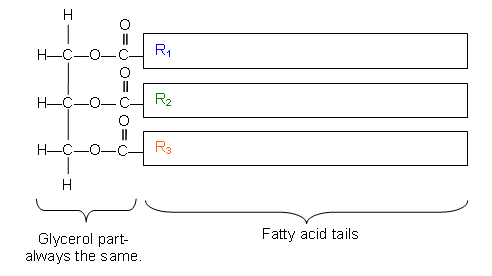
Fats and Fatty Acids
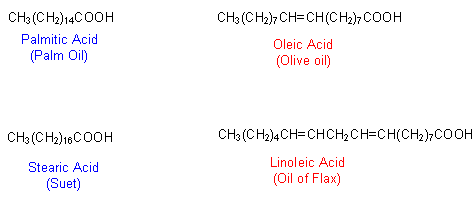
- The carboxylic acids in fats and oils are usually unbranched, containing an even number of carbon atoms ranging from C4 to C24.
- The fatty acids can either be fully saturated (all single bonds) or they can be unsaturated (contain one or more carbon-carbon double bond).
- Some common fatty acids are shown below:
Solid or Liquid?
- Natural oils and fats contain a mixture of many triesters.
- The nature of those fatty acids determines the properties of the substance and whether it is solid or liquid at room temperature.
- The following structure shows a saturated triglyceride:
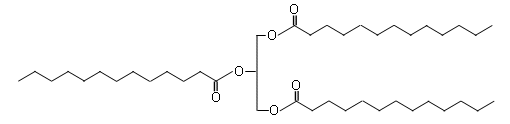
- As the chains are unbranched, the triglycerides can pack closely together; this means that the intermolecular forces holding the molecules together are stronger and therefore more energy is required to separate the molecules; hence it is solid at room temperature.
- Many doctors believe that saturated fats cause blockages within blood vessels and can therefore lead to heart disease.
- The following diagram shows the structure of an unsaturated triglyceride:
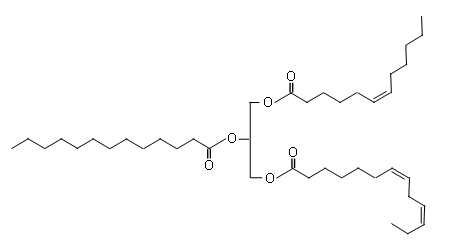
- The unsaturated triglyceride molecules can not pack so closely together due to the kink caused by the presence of the cis double bonds; the intermolecular forces are therefore weaker and so less energy is needed to separate the molecules, so they have a lower melting point and are liquid at room temperature.
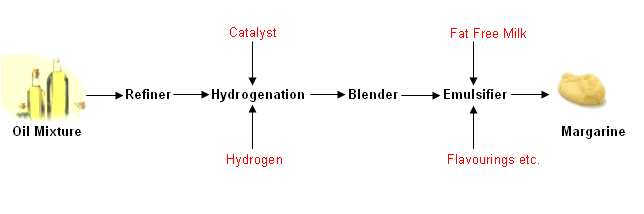
- Oils can be converted to fat in a hydrogenation reaction; this is how margarine is produced.
- The addition of hydrogen to unsaturated molecules reduces the number of double bonds; the triglycerides become more saturated.
- The conditions required for hydrogenation are a temperature of 180oC and a pressure 5 atmospheres using a nickel catalyst:
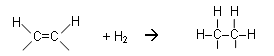
- This is an example of an addition reaction.
- It must be closely monitored, as if too much hydrogen is added, the fat can become too hard and brittle.
- The following flow diagram gives a simplified representation of how margarine is produced from oil:
Hydrolysis of Fats
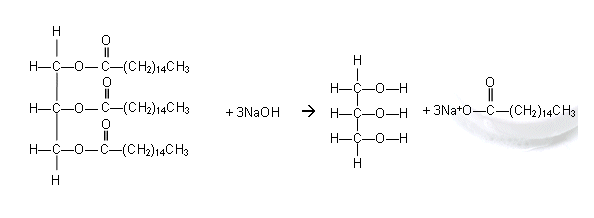
- Like all esters, the ester linkage holding the molecules together can be hydrolysed.
- When this is done with concentrated sodium hydroxide solution, a soap is made.
- Soaps are the sodium or potassium salts of fatty acids and they are made by heating the oils and fats with sodium or potassium hydroxide.
- The free fatty acids are then released from the sodium salts by adding a dilute acid.
Different Types of Oil
- There are different types of oil that can be used as mediums within paints.
- There are drying oils; when exposed to air, these become rubbery solids.
- There are non-drying oils, which do not dry, but can become hardened by heating.
- There are semi-drying oils which thicken and form a skin when exposed to air and high temperatures.
- Paint layers dry from the outside inwards to give a tacky surface first and eventually they become hard throughout. The drying process is irreversible.
Iodine Numbers
-
Natural oils can be classified according to their iodine number, which is based on how much iodine chloride (ICl) will react with the oil.
- The more ICl that reacts with the oil sample, the higher the iodine number.
- The higher the iodine number, the greater the drying potential the oil has.
- This can be related to the reaction that occurs between oils and iodine chloride; unsaturated bonds within the oil molecule can go through an electrophilic addition reaction with iodine chloride:
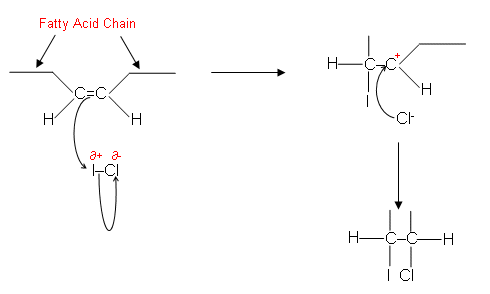
- This electrophilic addition reaction can only occur across unsaturated carbon carbon double bonds.
- The degree of saturation is also responsible for the drying potential of the oil; the more unsaturated the oil, the greater its drying potential.
- This is because in oxygen, a free radical polymerisation reaction occurs between the carbon carbon double bonds in adjacent molecules, cross-linking them together.
- This results in the hard protective coating being formed.
Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)
Salters (OCR) Revise A2 Chemistry Home
Home