
Monastral Blue
- Monastral blue is one of the most effective blue pigments ever made; it is used to colour plastics, in printing inks, paints and enamel because of its pure blue colour.
- It was discovered accidentally by Scottish Dyers in 1928; the iron from a reaction vessel contaminated the reaction mixture for the production of phthalamide (used for manufacture of dyes) producing a pure blue solution.
- The new compound synthesised was named iron phthylocyanine:
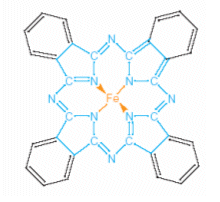
- Iron phthylocyanine is a large flat molecule, containing a 16-membered ring of alternating carbon and nitrogen atoms, with an iron atom at the centre.
- Four of the nitrogen atoms act as ligands to form a planar complex with the iron atom; complexes of transition metals are usually coloured (see Steel Story).
- The 16-membered ring in iron phthylocyanine contains alternating single and double bonds; this arrangement is called a conjugate system of bonds.
- For each double bond, a pair of electrons is not confined to bonding two particular carbon atoms (as in a single bond), but is delocalised over the whole conjugated system.
- The lone pair of electrons on the nitrogen atoms are also involved, and so the delocalisation spreads over the whole 16-membered ring; the delocalisation also spreads over the benzene rings.
- Delocalised electrons lead to a more stable structure; therefore iron phthylocyanine is a very stable pigment.
- Molecules with extended conjugated systems tend to be coloured and so they make ideal dyes and pigments.
Other Colours
- Once the structure of iron phthylocyanine had been discovered, a team of chemists at imperial college set to work on researching other phthylocyanines; replacing iron with other d-block transition metals.
- Within a few years, the team found that a whole range of similar pigments could be made.
- In 1939, copper phthylocyanine was on the market with the trade name Monastral Blue.
- The commercial importance of Monastral Blue and other phthylocyanine pigments is due to a combination of factors:
- They have beautiful bright blue to green shades with high colour strengths (meaning less pigment has to be used).
- They are very stable and have excellent fastness to light and so don’t fade.
Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)
Salters (OCR) Revise A2 Chemistry Home
Home


