
Gas-Liquid Chromatography
- Chromatography is an important analytical technique used by chemists to separate and identify the compounds in a mixture.
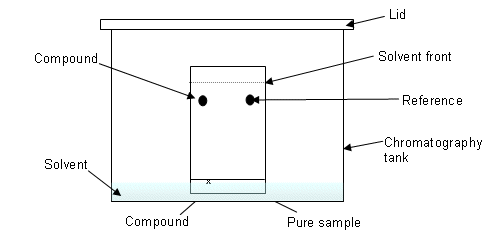
- There are a number of different types of chromatography, in What’s in a Medicine, thin layer chromatography was covered.
- All the different types of chromatography rely on the equilibrium established when a compound distributes itself between two phases; one mobile and the other stationary.
- Different compounds distribute themselves to different extents and so move along with the mobile phase at different speeds.
- In thin layer chromatography, the stationary phase is the solid support material (often the plastic chromatography plate). The mobile phase is the solvent that rises up the plate.
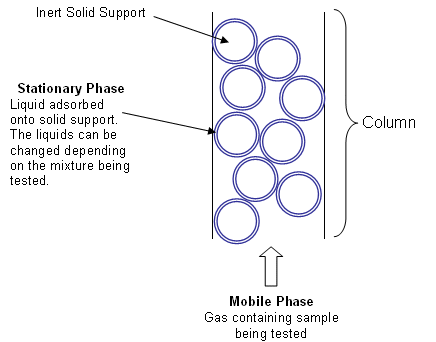
- In gas-liquid chromatography, the principle is the same, but the mobile phase is an unreactive gas, such as nitrogen (the carrier gas), and the stationary phase comprises of a small amount of liquid held on a finely-divided inert solid support. The solid support is in the form of a powder which is packed into a long, thin tube (the column).
- The column is coiled inside an oven.
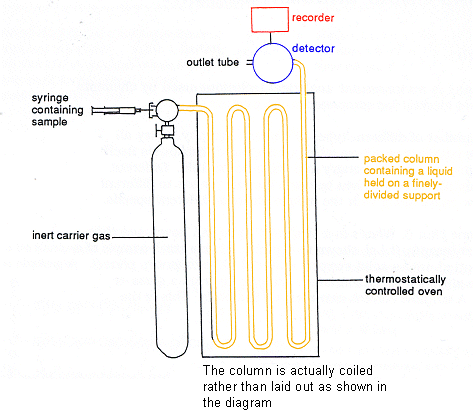
The Instrument
- The sample to be reacted is injected into the gas stream just before it enters the column.
- The components of the mixture are then carried through the column in a stream of gas.
- Each compound distributes itself between the phases to different extents and therefore emerges from the column at a different time.
- Some of the compounds dissolve in the stationary solvents more readily than others; these travel through the column slower and so emerge last.
- The most volatile compounds usually emerge first.
- A detector on the outlet tube monitors compounds emerging from the column.
- Signals from the detector are plotted out by a recorder as a chromatogram.
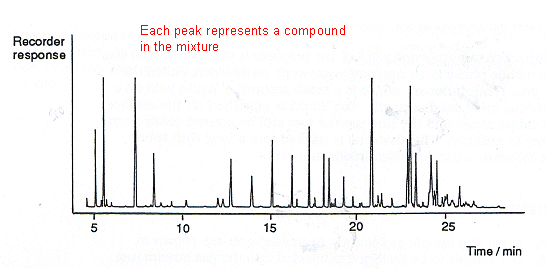
- The chromatogram shows the recorder response against the time which has elapsed since the sample was injected into the column.
- Each component of the mixture gives rise to a peak on the chromatogram.
- The following shows a gas chromatogram of a premium grade petrol:
- The time a compound is held on a column under given conditions is characteristic of each compound and is referred to as its retention time; this can be affected by many factors, such as:
- The length and packing of the column.
- The nature and flow rate of the carrier gas.
- The temperature of the column.
- The retention time can be used to identify the different compounds.
- The instrument is calibrated with known compounds, so that the conditions are kept constant throughout the analysis.
- The area under each peak represents the amount of each compound present.
- Gas-liquid chromatography is very sensitive and can be used to detect small quantities of substances; it is often used in forensic tests.
- In more sophisticated instruments, the outlet tube is connected to a mass spectrometer, so that each compound can be detected directly.
Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)
Salters (OCR) Revise A2 Chemistry Home
Home






