
The arenes
The Structure of Benzene
- Benzene is a colourless liquid with the molecular formula C6H6.
- As it has an equal number of carbon atoms to hydrogen atoms, so it must be very unsaturated; however it does not behave like other unsaturated molecules; it is very unreactive and has its own characteristic properties.
- The reason for this is related to the cyclic structure of benzene:
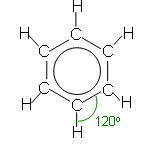
- The benzene ring is a flat hexagon, all of the bond angles are 120o and the carbon-carbon bonds are all the same length (0.139 nm)- the length is less than a carbon carbon double bond (0.154 nm), but more than a carbon carbon single bond (0.134).
- The structure of benzene puzzled scientists in the nineteenth century; they knew that its molecular formula was C6H6, but they could not work out how this could be.
- In 1856, August Kekulé proposed that benzene’s structure was cyclic and comprised of alternating single and double bonds:
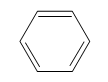
- However, this was disproved for two reasons; firstly all of the carbon carbon double bonds were found to be the same length; this can not be the case for double and single bonds. Secondly, the benzene ring is much more stable than the Kekulé structure suggests it should be.
- Rather than the alternating single and double bonds, the structure contains a delocalised electron system.
- Each carbon atom has four outer electrons which can be used to form bonds; three of these are used to form sigma bonds with the two adjacent carbon atoms and a hydrogen atom.
- The extra electrons, instead of overlapping in pairs to form pi bonds, are spread out evenly and are shared by all 6 carbons in the benzene ring (electron delocalisation).
- Additional evidence for the delocalised structure came from electron density maps which can be produced from x-ray diffraction studies.
Stability of Benzene
- The delocalised electrons in the benzene ring result in it being much more stable than it would be expected to be if it had the Kekulé type structure.
- The delocalisation allows the electrons in the molecule to be further apart. As electrons repel one another, the further apart they are, the lower the repulsion and therefore the more stable the structure.
- The stability of the benzene ring affects the reactions it can undergo; it will tend to only undergo reactions in which the stable ring is preserved.
- Those reactions which disrupt the delocalised electron system are less favourable and so require higher temperatures and more vigorous conditions.
- Thermodynamic experiments proved that the benzene ring did not have the proposed Kekulé structure; the enthalpy change when benzene reacts with hydrogen to form cyclohexane can be used to determine its stability:
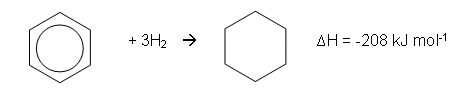
- A reasonable estimate can be determined for the reaction if benzene had the Kekulé structure.
- The enthalpy change for the reaction of cylcohexene with hydrogen to form cyclohexane is -120 kJ mol-1:
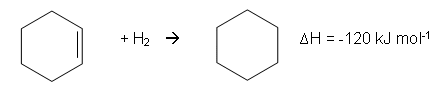
- Therefore, the enthalpy change for the Kekulé structure would be:
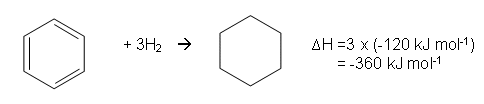
- This means that less energy is given out than would be expected if the structure had the Kekulé structure. As less energy has been given out, more energy has been used to break the bonds in the structure in order to put hydrogen across them, therefore the structure must be more stable.
The Arenes
- Hydrocarbons that contain rings stabilised by delocalised electrons, like benzene, are called arenes. The suffix –ene, refers to them being unsaturated, like the alkenes, and the prefix ar- comes from many of them having an aromatic fragrance.
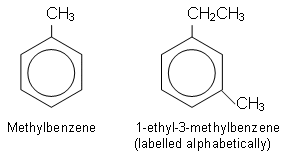
- There are many arenes, for example:
- Benzene rings can also join together to give fused ring systems, such as naphthalene and anthracene:
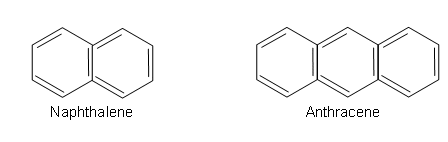
- The delocalised electrons are spread over all of the rings.
Arene Derivatives
- There are many derivatives of the benzene ring which are formed by replacing a hydrogen atom with different functional groups, for example:
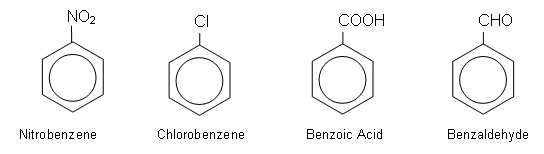
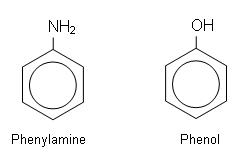
- The C6H5 group derived from benzene is referred to as the phenyl group.
- Two important aromatic compounds based on this group are phenol and phenylamine:
Useful books for revision
Revise A2 Chemistry for Salters (OCR A Level Chemistry B)
Salters (OCR) Revise A2 Chemistry Home
Home











